Preclinical & Translational Research
Saving Horseshoe Crabs, Advancing Science with Modern Alternatives for Pyrogen and Endotoxin Testing
In a landmark shift that blends scientific progress with environmental responsibility, the United States Pharmacopeia (USP) has endorsed the use of non-animal-derived reagents for endotoxin testing. This decision marks a pivotal advancement in pharmaceutical safety testing while paving the way for a more ethical and sustainable industry standard. For decades, the pharmaceutical and medical device sectors have relied on…
Read MoreThe Role of Human Skin Equivalent (HSE) Models in Advancing Monoclonal Antibody Drug Development
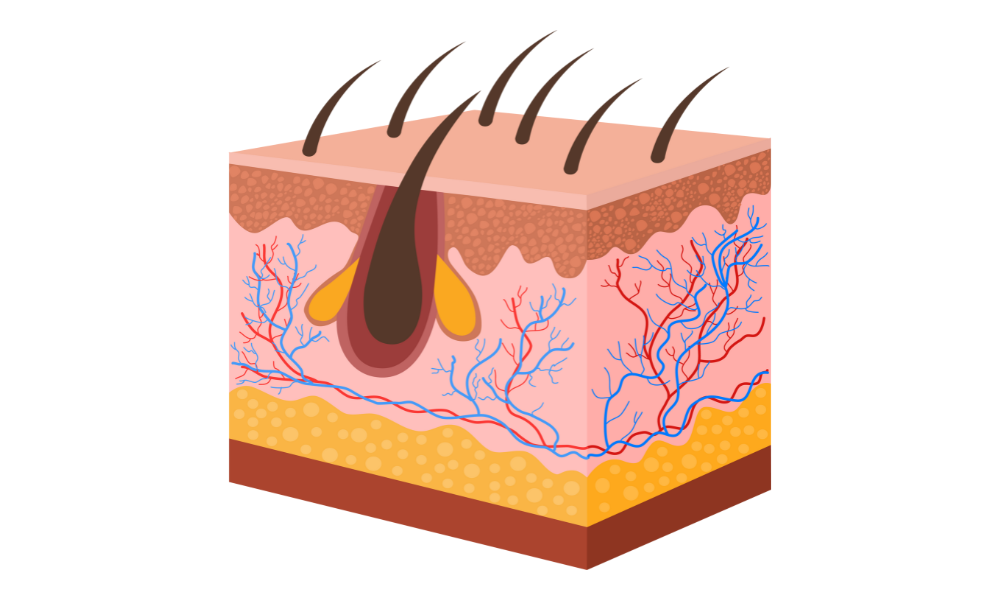
Monoclonal antibody (mAb) drug development is rapidly evolving, driven by growing demand for precision therapies and a regulatory push toward non-animal testing methods. In this dynamic environment, Human Skin Equivalent (HSE) models are emerging as a powerful preclinical tool, especially for mAbs targeting dermatologic conditions or systemic diseases with skin involvement. As the FDA moves to phase out…
Read MoreThe Importance of Viral Clearance Testing in Biotherapeutic Product Development
Viral clearance testing is a complex process that is crucial for ensuring the safety and efficacy of biotherapeutic products via demonstrating effective inactivation or removal of viruses during the manufacturing processing steps. Since each biotherapeutic product is unique in material, design, and processing, each viral clearance study is a custom design, but all have common…
Read MoreCytotoxicity and Cell Viability: The Big Picture
Determining cytotoxicity is a critical step in assessing the biocompatibility of medical devices, biomaterials, and drug screening. There are a wide variety of methods available to researchers; however, too often cell-based assay results indicating poor cell health without greater context slow down or halt progress on the development of a medical device. Technology is constantly…
Read MoreMedical Device Testing Strategies for an Effective Regimen
All medical device manufacturers strive to ensure devices and biomedical technologies are safe, efficacious, and fit for intended use. The biggest roadblock to accomplishing this is medical device testing to determine the most appropriate regimen for the device and its use. Although speed to market is key, a medical device CRO must devise and implement…
Read MoreEx Vivo Model Publication
We’d like to share the recent publication in Wound Management & Prevention by the great team at Rochal Industries Inc and iFyber team led by Nina Bionda. It highlights the efficacy of wound cleansers in two relevant biofilm models. The ex vivo porcine dermal model of mature biofilms is routinely used by our clients for the evaluation of…
Read MoreiFyber offers GLP compliant testing services
I’m pleased to announce that we have added GLP-compliant testing to our service offering. We focus on custom testing and method development early in the development process, especially for medical device, drug, and biologic manufacturers in the wound care and regenerative medicine space. GLP capability adds an additional level of quality review and assurance for…
Read MoreEx Vivo Biofilm Model: A Bridge to Animal Models
iFyber today released a whitepaper on an ex vivo model for mature biofilms. The model provides medical device and drug developers a new option to bridge the gap between traditional in vitro biofilm models and in vivo animal studies. Read more here.
Read More