Microbiology & Antimicrobial Testing
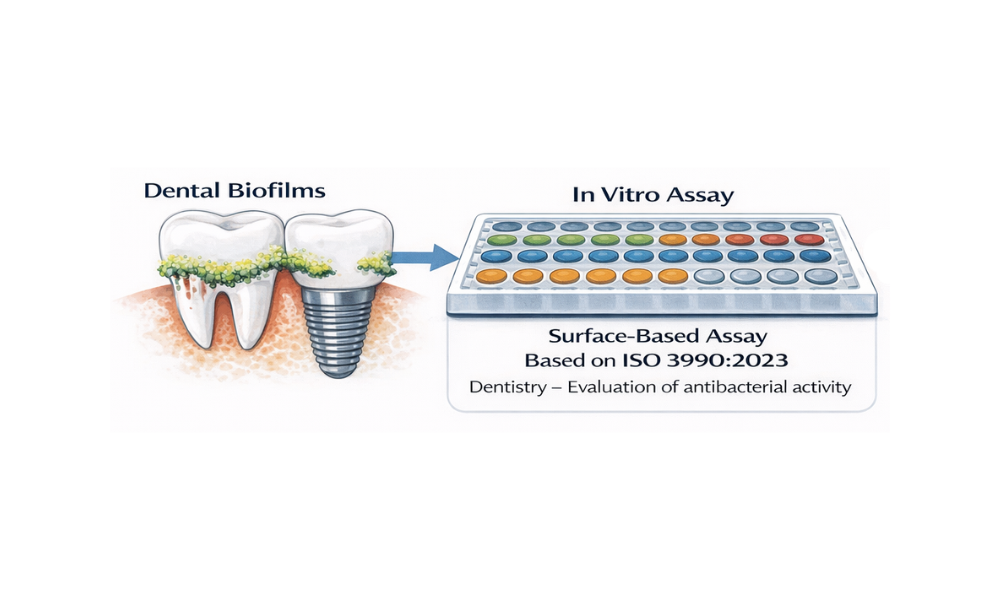
Advancing Dental Material Biofilm Testing with High-Throughput In Vitro Assays
For teams developing dental and implant materials, biofilm performance is a defining factor in clinical success, regulatory acceptance, and product differentiation. The challenge is not in understanding biofilms, but generating reliable, clinically relevant data that reflects how microbial communities interact with material surfaces under real-world conditions. Traditional antimicrobial testing approaches often fall short in this context. To…
Read MoreManaging Antimicrobial Resistance in Chronic Wound Care
Slow-healing wounds, like diabetic foot ulcers, venous leg wounds, and pressure ulcers, frequently turn into long-term battles. When dealing with chronic wounds, treatment can be difficult to target due to a multitude of complicating factors. Diabetes, vascular disease, and immune suppression all reduce the body’s ability to fight infection and repair tissue. Additionally, microbes within…
Read MoreBiofilm Disruptors in Wound Care Products: Moving Beyond Antimicrobials
Since the 1940s, antimicrobials have been our first line of defense in wound care. However, the very thing that has helped us fight infections is now starting to help the infections fight back. Structured communities of microorganisms, like bacteria, fungi, or yeast—called biofilms— are significantly more tolerant to antimicrobial treatments and harder to eradicate than…
Read MoreBiofilm Testing: The Critical Step in Advancing Infection-Resistant Medical Devices
Infections related to medical devices remain a persistent and costly challenge in modern healthcare. Despite the advancement of antimicrobial materials and design innovations, biofilm formation continues to undermine the efficacy of many devices – contributing to patient complications, prolonged hospital stays, and increased clinician workload. Biofilm formation on medical devices remains a major barrier to infection prevention.…
Read MoreISO and USP Standards for Antimicrobial Effectiveness Testing: What You Need to Know
From toothbrushes to tongue scrapers, oral care products tackle one of the most microbiologically active environments in the human body, with conditions where diverse microbial species thrive. This makes antimicrobial effectiveness testing essential, and proves to be a unique challenge for product developers: proving their antimicrobial claims while satisfying strict regulatory standards. USP and ISO…
Read MoreOral Care Innovation: Delivering Antimicrobial Solutions Without Compromising Safety
The mouth is an active place. At any given moment, there are billions of bacteria alive and multiplying. While many are beneficial, others pose more serious risks. As medical understanding of the oral environment and its microbial communities advances, innovative oral care products are being developed that focus on safe, targeted antimicrobial solutions. However, developing…
Read MoreBeyond Pass/Fail: Why USP 51 Demands a More Scientific Approach to Antimicrobial Efficacy Testing
Pharmaceutical, cosmetic, and personal care products containing aqueous components are susceptible to microbial contamination—especially during manufacturing and consumer use. To ensure product safety and stability over time, the United States Pharmacopeia (USP) <51> Antimicrobial Effectiveness Testing (AET) remains one of the most widely accepted standardized methods for evaluating preservative performance. However, many testing providers offer…
Read MoreLooking for a Reliable Biofilm Testing Solution?
Experience Advanced Support with a Trusted Preclinical CRO Partner. Biofilms are complex communities of microorganisms that can adhere to surfaces, forming structures that can potentially cause infections and pose significant risks in healthcare settings. Their resistance to antimicrobial agents brings an added layer of complexity. Understanding biofilm formation and its impact during the development of pharmaceuticals and medical…
Read MoreScience Meets Smile: Evaluating the Effectiveness of Antimicrobial Dental Products
Dental products come into contact with a diverse array of microorganisms. With over 700 microbial species identified in this environment, it’s a complex ecosystem to navigate. Fahimeh Tabatabaei, a dentist with a PhD in dental biomaterials, explains that not all bacteria residing in the oral cavity pose a threat. ” Some of these microorganisms form…
Read MoreEvaluation of Antimicrobial Efficacy
Antimicrobial efficacy testing is crucial for assessing the performance and safety of antimicrobial solutions and products used in clinical settings. These tests evaluate the products’ ability to prevent or inhibit the growth of microorganisms, reducing the risk of healthcare-associated infections (HAIs) and ensuring patient safety. They provide a systematic approach to assess the products’ effectiveness…
Read More